Xtandi Fact Sheet
Why are we here?
1. Xtandi is an important cancer drug that improves quality of life for a large patient population
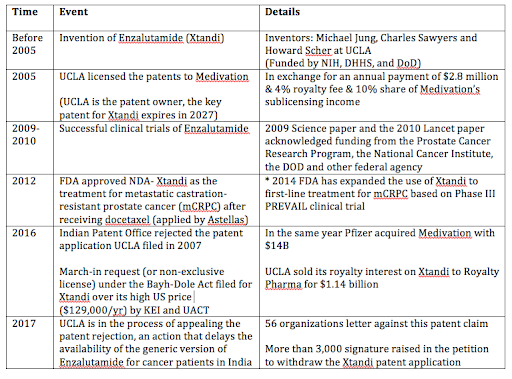
2. Xtandi was developed at UCLA with federal funding from its creation to its clinical trials.
3. Pharmaceutical companies are exploiting its monopoly power to set Xtandi at a prohibitive price and are attempting to so in India at the cost of restricting patient access
4. UC appears complicit with Pharma’s disregards to patient access and contrasting with our licensing guideline in its Xtandi patent pursuit in India.
5. UC is the number one university in drug invention with a sincere interest in promoting global health. What are our roles and tools to counter-balance the malicious price hike in drugs we created such as Xtandi?
6. There is an increasing public awareness of Xtandi and US distorted drug price. UCLA can be a role model by adopting a responsible licensing practice that prohibits Pharma’s exploitation of patent rights.
What would we like to propose?
1. Explore our roles, responsibilities, and power in bringing drugs to market at reasonable prices
2. Issue public statement on our stance on Xtandi
3. Issue public statement on our re-commitment in responsible licensing that protects patient access
4. Do the right thing for our patent decision in India
UCLA licensing guideline and its endorsement of “Nine points to consider”:
Universities have a social compact with society. As educational and research institutions, it is our responsibility to generate and transmit knowledge, both to our students and the wider society. We have a specific and central role in helping to advance knowledge in many fields and to manage the deployment of resulting innovations for the public benefit. In no field is the importance of doing so clearer than it is in medicine. Around the world, millions of people are suffering and dying from preventable or curable diseases. The failure to prevent or treat disease has many causes. We have a responsibility to try to alleviate it, including finding a way to share the fruits of what we learn globally, at sustainable and affordable prices, for the benefit of the world’s poor. There is an increased awareness that responsible licensing includes consideration of the needs of people in developing countries and members of other underserved populations.
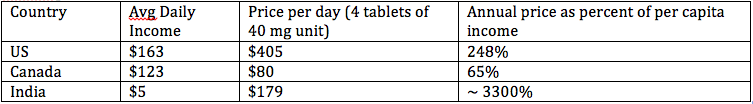
Xtandi price:
* Recommended dosage of Xtandi is 4 tablets a day. Average course of treatment is 8 months
Xtandi cost & sales:
Cost of production: $303 million (according to Medivation SEC filing, R&D outlays on Xtandi from 2005-2012)
Sales:
$5 billion from the US market alone through Sept 10th 2018 with billions more from sales outside of the US, based on Astella's earning report.
Xtandi market:
Globally, it is the second most common type of cancer and the fifth leading cause of cancer-related death in men. (In 2012, it occurred in 1.1 million men and caused 307,000 deaths)
When patients are treated early and tumors are localized, the prognosis is often favorable. However, some patients will relapse, leading in nearly all cases to CRPC. Xtandi is found to be effective and improves quality of life for patients with CRPC. Xtandi/Enzalutamide is also being tested for other types of cancer (breast cancer, hepatocellular carcinoma, bladder cancer, ovarian cancer, pancreatic cancer, mantle cell lymphoma).
The number of patients receiving Xtandi is increasing:
2,143 in 2012, 7,329 in 2013, 11,800 in 2014.
Why are we here?
1. Xtandi is an important cancer drug that improves quality of life for a large patient population
2. Xtandi was developed at UCLA with federal funding from its creation to its clinical trials.
3. Pharmaceutical companies are exploiting its monopoly power to set Xtandi at a prohibitive price and are attempting to so in India at the cost of restricting patient access
4. UC appears complicit with Pharma’s disregards to patient access and contrasting with our licensing guideline in its Xtandi patent pursuit in India.
5. UC is the number one university in drug invention with a sincere interest in promoting global health. What are our roles and tools to counter-balance the malicious price hike in drugs we created such as Xtandi?
6. There is an increasing public awareness of Xtandi and US distorted drug price. UCLA can be a role model by adopting a responsible licensing practice that prohibits Pharma’s exploitation of patent rights.
What would we like to propose?
1. Explore our roles, responsibilities, and power in bringing drugs to market at reasonable prices
2. Issue public statement on our stance on Xtandi
3. Issue public statement on our re-commitment in responsible licensing that protects patient access
4. Do the right thing for our patent decision in India
UCLA licensing guideline and its endorsement of “Nine points to consider”:
Universities have a social compact with society. As educational and research institutions, it is our responsibility to generate and transmit knowledge, both to our students and the wider society. We have a specific and central role in helping to advance knowledge in many fields and to manage the deployment of resulting innovations for the public benefit. In no field is the importance of doing so clearer than it is in medicine. Around the world, millions of people are suffering and dying from preventable or curable diseases. The failure to prevent or treat disease has many causes. We have a responsibility to try to alleviate it, including finding a way to share the fruits of what we learn globally, at sustainable and affordable prices, for the benefit of the world’s poor. There is an increased awareness that responsible licensing includes consideration of the needs of people in developing countries and members of other underserved populations.
Xtandi price:
* Recommended dosage of Xtandi is 4 tablets a day. Average course of treatment is 8 months
Xtandi cost & sales:
Cost of production: $303 million (according to Medivation SEC filing, R&D outlays on Xtandi from 2005-2012)
Sales:
$5 billion from the US market alone through Sept 10th 2018 with billions more from sales outside of the US, based on Astella's earning report.
Xtandi market:
Globally, it is the second most common type of cancer and the fifth leading cause of cancer-related death in men. (In 2012, it occurred in 1.1 million men and caused 307,000 deaths)
When patients are treated early and tumors are localized, the prognosis is often favorable. However, some patients will relapse, leading in nearly all cases to CRPC. Xtandi is found to be effective and improves quality of life for patients with CRPC. Xtandi/Enzalutamide is also being tested for other types of cancer (breast cancer, hepatocellular carcinoma, bladder cancer, ovarian cancer, pancreatic cancer, mantle cell lymphoma).
The number of patients receiving Xtandi is increasing:
2,143 in 2012, 7,329 in 2013, 11,800 in 2014.